Imagine waking up in the morning and checking your mobile phone. The phone works because of a battery. When you start your car, the engine begins running because of a battery. Even solar power systems and laptops depend on batteries to store and supply electrical energy.
Batteries are one of the most important parts of modern electrical systems. From small electronic devices to large backup power systems, batteries help store energy and provide electricity whenever it is needed. Without batteries, many technologies we use every day would not function properly.
Understanding battery types is very important for electrical students, engineers, technicians, and beginners. Different batteries have different characteristics, working principles, and applications. Choosing the correct battery type can improve efficiency, safety, and equipment performance.
In this article, you will learn the battery types, battery working principle, battery applications, and battery advantages and disadvantages. You will also understand how batteries work, their main components, and how to select the right battery for different electrical systems.
2. What is a Battery?
A battery is an electrical device that stores chemical energy and converts it into electrical energy. It provides power to electrical circuits and electronic devices.
In simple words, a battery acts like a small power station that stores energy and releases it when needed.
Inside a battery, chemical reactions take place between different materials. These reactions create an electrical current that flows through wires and powers devices.
Practical Example
For example, when you press the power button on a flashlight, the battery inside it starts supplying electricity to the bulb. This electrical energy produces light.
Similarly, mobile phones, laptops, cars, solar systems, and emergency backup systems all rely on batteries to operate.
3. Battery Working Principle
The battery working principle is based on a chemical reaction called an electrochemical reaction.
Inside the battery, chemicals react with each other to produce electrical energy.
Step-by-Step Working Principle
The working process of a battery can be explained in simple steps:
• A battery contains two electrodes: positive electrode (cathode) and negative electrode (anode).
• These electrodes are placed inside a chemical solution called electrolyte.
• A chemical reaction occurs between the electrodes and electrolyte.
• This reaction produces electrons at the negative terminal.
• Electrons move through the external circuit to the positive terminal.
• This movement of electrons creates electric current.
Simple Analogy
You can imagine a battery like a water tank.
The stored water represents stored energy, and when a tap is opened, water flows out.
Similarly, when a circuit is connected, stored chemical energy is converted into electrical energy.
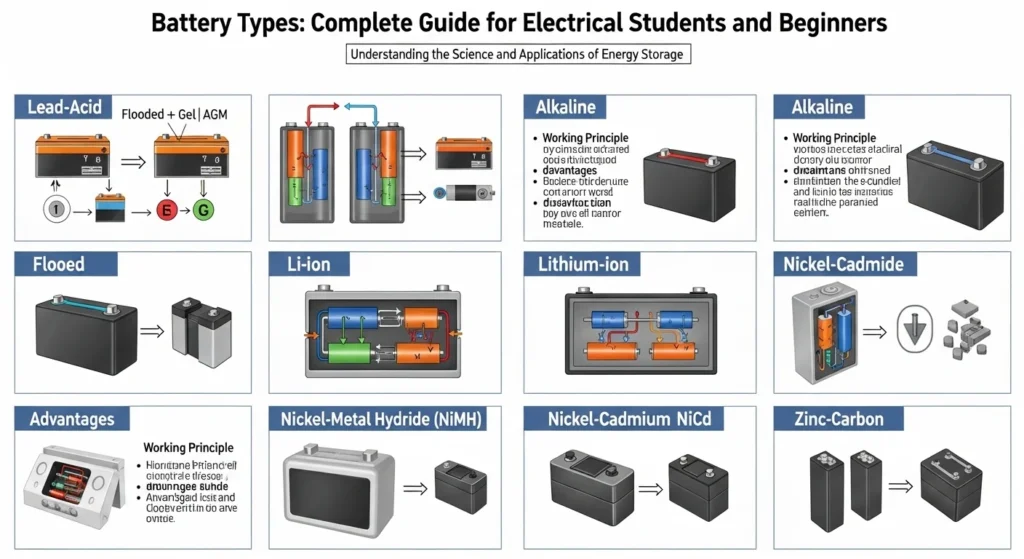
4. Types of Batteries
There are many battery types used in electrical systems. These batteries are generally divided into two main categories.
Primary Batteries
Primary batteries are non-rechargeable batteries. Once their energy is used, they cannot be recharged.
Common Examples
• Alkaline battery
• Zinc-carbon battery
• Lithium primary battery
Features
• Simple design
• Low cost
• Used in small devices
Applications
• Remote controls
• Wall clocks
• Flashlights
• Toys
Secondary Batteries
Secondary batteries are rechargeable batteries. They can be used many times by recharging them.
Examples
• Lead-acid battery
• Lithium-ion battery
• Nickel-cadmium battery
• Nickel-metal hydride battery
Features
• Rechargeable
• Longer lifespan
• Suitable for heavy-duty applications
Applications
• Electric vehicles
• Solar power systems
• UPS systems
• Mobile phones
Lead-Acid Battery
Lead-acid batteries are one of the oldest and most widely used battery types.
Characteristics
• Uses lead plates and sulfuric acid electrolyte
• Low cost
• High current capability
Applications
• Car batteries
• Solar systems
• UPS backup power
Lithium-Ion Battery
Lithium-ion batteries are modern rechargeable batteries widely used in electronics.
Characteristics
• Lightweight
• High energy density
• Long life
Applications
• Smartphones
• Laptops
• Electric vehicles
• Power banks
Nickel-Cadmium Battery
Nickel-cadmium batteries are durable rechargeable batteries.
Characteristics
• Long life cycle
• Works well in extreme temperatures
Applications
• Power tools
• Emergency lighting
• Aviation systems
Nickel-Metal Hydride Battery
This battery type improved upon nickel-cadmium technology.
Characteristics
• Higher capacity
• More environmentally friendly
Applications
• Hybrid cars
• Digital cameras
• Rechargeable household batteries
5. Main Components of a Battery
A battery contains several important parts that work together.
Electrodes
Electrodes are the positive and negative terminals where chemical reactions occur.
Electrolyte
Electrolyte is the chemical liquid or gel that allows ions to move inside the battery.
Separator
A separator prevents the electrodes from touching each other while allowing ion flow.
Battery Case
The battery case protects internal components and prevents leakage.
Terminals
Terminals connect the battery to electrical circuits.
6. Battery Advantages
There are many battery advantages and disadvantages, and batteries offer several important benefits.
Advantages
• Portable power source
• Provides electricity without direct power supply
• Essential for backup power systems
• Useful in renewable energy systems
• Supports mobile and portable devices
• Reliable for emergency power
Batteries allow electrical devices to operate anywhere, even without grid electricity.
7. Battery Disadvantages / Limitations
Despite their benefits, batteries also have some limitations.
Disadvantages
• Limited lifespan
• Requires charging or replacement
• Can be expensive for large systems
• Performance decreases over time
• Some batteries contain toxic materials
Proper handling and recycling are necessary to reduce environmental impact.
8. Battery Applications
Battery applications are found in almost every modern electrical system.
Home Applications
• Remote controls
• Flashlights
• Laptops
• Mobile phones
• Solar backup systems
Industrial Applications
• UPS power systems
• Electric forklifts
• Backup generators
• Communication systems
Modern Technology Applications
• Electric vehicles
• Renewable energy storage
• Smart devices
• Medical equipment
The growth of renewable energy is increasing the demand for advanced battery technology.
9. Comparison of Battery Types
Understanding the difference between battery types helps in selecting the right battery.
| Battery Type | Rechargeable | Cost | Lifespan | Common Use |
|---|---|---|---|---|
| Alkaline | No | Low | Short | Remote controls |
| Lead-Acid | Yes | Low | Medium | Cars, solar systems |
| Lithium-Ion | Yes | High | Long | Smartphones, EVs |
| Nickel-Cadmium | Yes | Medium | Long | Power tools |
| Nickel-Metal Hydride | Yes | Medium | Medium | Cameras |
Each battery type has advantages depending on the application.
10. Selection Guide: How to Choose the Right Battery
Selecting the right battery depends on several factors.
Capacity
Choose a battery with the correct ampere-hour (Ah) rating.
Voltage
Ensure the battery voltage matches the device requirements.
Lifespan
Some batteries last longer than others.
Cost
Budget is an important factor when choosing batteries.
Safety
Use reliable and certified battery products.
For beginners, lithium-ion batteries are common for electronics, while lead-acid batteries are widely used for backup power systems.
11. Common Battery Problems & Solutions
Problem 1: Battery Not Charging
Possible Causes:
• Faulty charger
• Damaged battery
Solution:
Check charger and battery connections.
Problem 2: Battery Drains Quickly
Possible Causes:
• Old battery
• High power consumption
Solution:
Replace the battery or reduce load.
Problem 3: Battery Overheating
Possible Causes:
• Overcharging
• Poor ventilation
Solution:
Use proper chargers and avoid overcharging.
12. Future Trends in Battery Technology
Battery technology is improving rapidly due to the growing demand for renewable energy and electric vehicles.
Solid-State Batteries
These batteries promise higher safety and energy density.
Fast-Charging Technology
New batteries are being designed to charge much faster.
Renewable Energy Storage
Advanced batteries will play a key role in storing solar and wind energy.
Electric Vehicle Batteries
Research is focused on increasing battery range and reducing charging time.
In the future, batteries will become safer, more efficient, and more environmentally friendly.
13. Conclusion
Batteries are essential components of modern electrical systems. They store energy and provide electricity to devices ranging from small electronics to large power systems. Understanding different battery types helps electrical students, engineers, and technicians choose the right power source for their applications.
In this article, we explored the battery working principle, battery applications, battery advantages and disadvantages, and the main battery classifications. We also discussed how batteries work, their components, and how to select the right battery for different uses.
As technology continues to evolve, batteries will play an even greater role in renewable energy, electric vehicles, and portable electronics. For electrical professionals and students, learning about battery technology is an important step toward understanding modern power systems.