Imagine your smartphone battery running out during an important call. You simply plug the charger into a power outlet, wait for some time, and the battery becomes ready to use again. The same happens with laptops, electric vehicles, UPS systems, and many other devices. The reason this is possible is because these devices use secondary batteries, also known as rechargeable batteries.
Secondary batteries have become one of the most important technologies in modern electrical and electronic systems. Unlike disposable batteries, they can be charged and used multiple times, making them more economical and environmentally friendly.
Understanding secondary batteries is essential for electrical students, engineers, technicians, and beginners who want to learn how modern power storage systems work. These batteries play a critical role in renewable energy systems, electric vehicles, portable electronics, and backup power systems.
In this article, you will learn about the secondary batteries working principle, secondary batteries applications, and secondary batteries advantages and disadvantages. We will also explore different types of secondary batteries, their components, how they work, and how to select the right battery for different electrical applications.
2. What are Secondary Batteries?
A secondary battery is a type of battery that can be recharged and reused many times by passing electrical current through it.
Simple Definition
Secondary batteries store electrical energy in chemical form and release it as electrical energy when required. Unlike primary batteries, they can be recharged after discharge.
Simple Explanation
Inside a secondary battery, chemical reactions occur between electrodes and electrolyte. These reactions store energy during charging and release energy during discharging.
Practical Example
A car battery is a common example of a secondary battery. When the engine runs, the alternator charges the battery. Later, the battery provides power to start the engine or operate electrical systems.
This rechargeable feature makes secondary batteries extremely useful in modern electrical systems.
3. Secondary Batteries Working Principle
The secondary batteries working principle is based on reversible electrochemical reactions.
During charging and discharging, chemical reactions occur between the battery electrodes and electrolyte.
Step-by-Step Working Process
During Discharging
• Chemical energy converts into electrical energy
• Electrons flow through the external circuit
• The device receives electrical power
During Charging
• External power source supplies electricity
• Electrical energy converts back into chemical energy
• The battery stores energy for future use
Simple Analogy
Think of a water tank.
• Filling the tank = Charging the battery
• Using water from the tank = Discharging the battery
The tank can be filled again and again, just like a rechargeable battery.
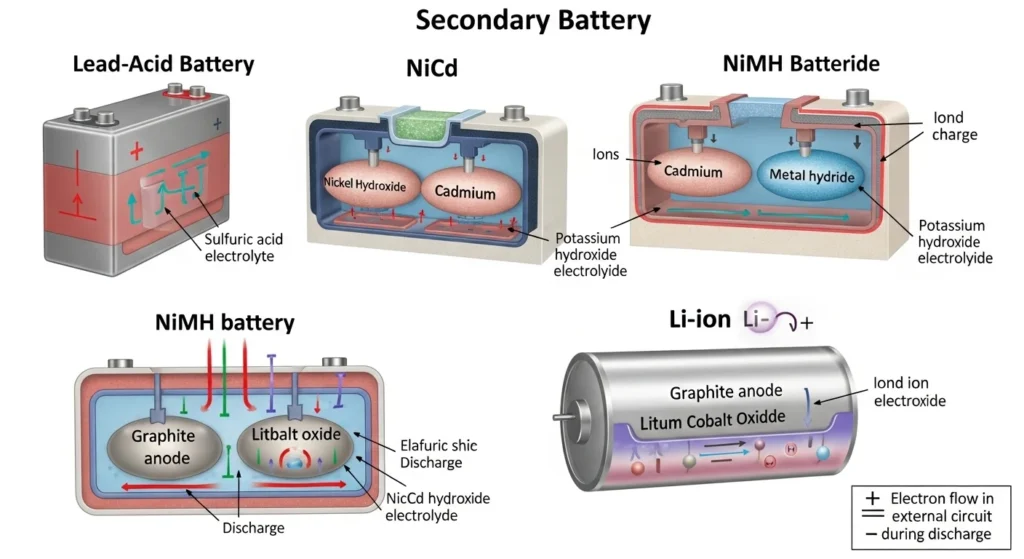
4. Types of Secondary Batteries
There are several types of secondary batteries used in electrical and electronic systems.
Lead-Acid Battery
The lead-acid battery is one of the oldest and most widely used secondary batteries.
Features
• Uses lead plates and sulfuric acid electrolyte
• Reliable and low-cost
• High current capability
Applications
• Automobile batteries
• UPS systems
• Backup power systems
Lead-acid batteries are very common in vehicles and emergency power systems.
Lithium-Ion Battery
Lithium-ion batteries are modern rechargeable batteries used in portable electronics.
Features
• High energy density
• Lightweight
• Long cycle life
Applications
• Smartphones
• Laptops
• Electric vehicles
• Power banks
Lithium-ion batteries are widely used because they provide high performance in compact size.
Nickel-Cadmium Battery (NiCd)
Nickel-cadmium batteries are durable rechargeable batteries.
Features
• Long cycle life
• High discharge capability
• Good performance in extreme temperatures
Applications
• Emergency lighting
• Aviation systems
• Industrial equipment
However, environmental concerns have reduced their use.
Nickel-Metal Hydride Battery (NiMH)
Nickel-metal hydride batteries are an improved version of NiCd batteries.
Features
• Higher capacity
• Less environmental impact
• Reliable performance
Applications
• Hybrid vehicles
• Rechargeable household batteries
• Electronic devices
NiMH batteries are widely used as replacements for older battery technologies.
5. Main Components of Secondary Batteries
Secondary batteries contain several important components that enable energy storage.
Electrodes
There are two electrodes inside the battery.
• Anode – Negative electrode
• Cathode – Positive electrode
They store and release electrical energy through chemical reactions.
Electrolyte
The electrolyte allows ions to move between electrodes during charging and discharging.
Separator
The separator prevents the electrodes from touching while allowing ion flow.
Battery Container
The container holds all battery components and protects them from external damage.
Terminals
Battery terminals provide connection points for electrical circuits.
6. Secondary Batteries Advantages
Understanding secondary batteries advantages and disadvantages helps engineers choose the right battery for applications.
Advantages
• Rechargeable and reusable
• Cost-effective over long-term use
• Environmentally friendly compared to disposable batteries
• Suitable for high power applications
• Widely available in different sizes and capacities
• Essential for renewable energy storage
These benefits make secondary batteries essential in modern technology.
7. Secondary Batteries Disadvantages / Limitations
Despite their advantages, secondary batteries also have some limitations.
Disadvantages
• Higher initial cost compared to primary batteries
• Require charging equipment
• Performance decreases over time
• Limited number of charge cycles
• Some types require maintenance
Engineers must consider these limitations when designing battery systems.
8. Secondary Batteries Applications
Secondary batteries have many important applications in modern electrical systems.
Home Applications
• Smartphones
• Laptops
• Tablets
• Portable electronic devices
Automotive Applications
• Car starter batteries
• Electric vehicles
• Hybrid vehicles
Industrial Applications
• UPS systems
• Backup power supply
• Telecom power systems
Renewable Energy Applications
• Solar energy storage
• Wind energy storage
• Off-grid power systems
These applications make secondary batteries a key part of modern power infrastructure.
9. Comparison: Secondary Batteries vs Primary Batteries
Understanding the difference between primary batteries and secondary batteries helps beginners understand battery technologies.
| Feature | Secondary Batteries | Primary Batteries |
|---|---|---|
| Rechargeable | Yes | No |
| Lifespan | Long | Short |
| Cost | Higher initial cost | Lower initial cost |
| Environmental Impact | Lower | Higher |
| Applications | Electronics, vehicles | Remote devices |
Secondary batteries are more suitable for repeated use and high-energy applications.
10. Selection Guide: How to Choose Secondary Batteries
Choosing the correct secondary battery depends on several important factors.
Energy Capacity
Select a battery with sufficient capacity for the device.
Battery Type
Choose between lead-acid, lithium-ion, or NiMH based on application.
Cycle Life
Higher cycle life batteries last longer.
Charging Requirements
Ensure compatible charging systems are available.
Operating Environment
Temperature and environmental conditions affect battery performance.
Proper selection improves efficiency and battery lifespan.
11. Common Problems & Solutions
Problem 1: Battery Not Charging
Possible Causes
• Faulty charger
• Damaged battery
Solution
Check charger connections or replace the battery.
Problem 2: Battery Drains Quickly
Possible Causes
• Aging battery
• High power consumption
Solution
Replace the battery or reduce load.
Problem 3: Overheating Battery
Possible Causes
• Overcharging
• Internal short circuit
Solution
Use proper charging equipment and avoid overcharging.
12. Future Trends in Secondary Batteries
Battery technology is rapidly evolving.
Solid-State Batteries
Future batteries may use solid electrolytes for improved safety.
Advanced Lithium Batteries
Higher energy density and faster charging capabilities.
Renewable Energy Storage
Secondary batteries will play a major role in solar and wind energy systems.
Electric Vehicle Growth
The demand for high-performance rechargeable batteries will continue to grow.
These innovations will shape the future of electrical energy storage.
13. Conclusion
Secondary batteries are an essential part of modern electrical and electronic systems. Their ability to store energy and be recharged many times makes them extremely valuable in today’s technology-driven world.
In this guide, we explored the secondary batteries working principle, secondary batteries applications, and secondary batteries advantages and disadvantages. We also discussed common battery types such as lead-acid, lithium-ion, nickel-cadmium, and nickel-metal hydride batteries.
Understanding the difference between primary batteries and secondary batteries helps electrical students and engineers select the right energy storage solution for different applications.
As renewable energy systems, electric vehicles, and portable electronics continue to expand, secondary batteries will become even more important. Learning about battery technologies is an important step for anyone pursuing knowledge in electrical engineering or modern power systems.